INTRODUCTION
Diabetes mellitus (DM), commonly known as diabetes, is a class of metabolic diseases characterized by hyperglycaemia (elevated level of glucose in the blood) (WHO, 2017), due to either the inability of the pancreas to produce enough insulin or the cells of the body are not responding properly to the produced insulin (Shoback, 2011). The prevalence of this menace is growing with increasing global population; about 300 million people were reported by World Health Organization (WHO) in 2010 to be afflicted with the disease, thus, becoming one of the major contributors to ill-health and premature mortality (WHO, 2013) and fifth leading cause of death globally (Alwan and Maclean, 2009).
The symptoms of diabetes may be acute, subacute, or chronic complications including frequent urination, increased thirst, increased hunger, diabetic ketoacidosis, non-ketotic hyperosmolar coma, cardiovascular disease, stroke, kidney failure, foot ulcers, retinopathy etc. (Kitabchi et al., 2009; WHO, 1999). Interestingly, in type 1 DM, there is a loss of insulin-producing beta cells of the islets of Langerhans in the pancreas, leading to insulin deficiency (Rother, 2007) while in the case of type 2 DM, insulin resistance may occur and may be combined with relatively reduced insulin secretion (Shoback, 2011). The other type of diabetes aside dementia resulting from obesity regarded as type 3 (Anstey et al., 2011; Nepal et al., 2014) is gestational diabetes mellitus though uncommon, but resembles type 2 diabetes in several respects, involving a combination of relatively inadequate insulin secretion and responsiveness (Willi et al., 2007). Above all, DM is associated with an increased risk of cardiovascular diseases mediated through oxidative stress (Diogo et al., 2013). Reactive oxygen species (ROS) such as hydroxyl radical, superoxide anion, nitrogen peroxide, etc. can directly damage lipids, carbohydrates, proteins, or deoxyribonucleic acid (Lobo et al., 2010; Young and Woodside, 2001) and modulate intracellular signaling pathways, such as mitogen-activated protein kinases and redox-sensitive transcription factors causing changes in protein expression with irreversible oxidative modifications (Yang et al., 2011). Hyperglycaemia-induced mitochondrial dysfunction and endoplasmic reticulum stress has been shown to promote ROS accumulation, accelerates cellular damage, and significantly contributes to the development and progression of diabetic complications (Folli et al., 2011).
Lessertia montana (Family: Fabaceae), formerly referred to as Sutherlandia montana (Goldblatt and Manning, 2000) and locally known as Mountain Balloon Pea, is a South African indigenous medicinal plant species with soft-wooded shrub, usually about 0.5–1.0 m high and characterized by a beautiful silvery green foliage, large red flowers, and attractive bladdery pods (van Wyk and Gericke, 2000). Indigenously, the infusion of the plant made from leaves is valued by the Basotho people of Eastern Free State, South Africa for sedative use as well as in the treatment of cardiac ailments (Moffett, 2010). However, the report of Alimi and Ashafa (2017) from our group on the antioxidant and antidiabetic activities in vitro provided an insight to the pharmacological uses of this plant on the leaves though the pharmacological potentials of the other parts of the plant remains extensively untapped. Moreover, it is worthy of mention the arrays of scientific evidences of a related species, Lessertia frutescens reported with anticancer, anti-stress, antidepressant, anti-HIV, antioxidant, antidiabetic, etc. activities on the leaf and whole plant in both in vitro and in vivo studies as reviewed by van Wyk and Albrecht (2008) and recently by Aboyade et al. (2014). In line with afore-mentioned, coupled with scarce information on other parts of L. montana in the literature, we decided to investigate further the antioxidative and antidiabetic implications of the seeds and pod. The present study was designed to determine the qualitative and quantitative phytochemical screening, as well as evaluate the antioxidant and antidiabetic activities of the extracts of these parts through the in vitro studies.
MATERIALS AND METHODS
Plant collection, identification, and authentication
Fresh L. montana containing all parts were plucked in November 2017 at Kestel town, Free State Province, South Africa. The proper identification and authentication of the plant were done by Dr. Ashafa AOT of the Department of Plant Sciences, University of the Free State (QwaQwa campus). A voucher specimen (AsfMed/01/2017/QHb) was raised and deposited at the departmental herbarium.
Extract preparation
The pod separated from the plant was rinsed with distilled water in order to remove dust particles and then opened to access the seeds. The pod and seed components were spread on a brown cardboard paper and exposed to air at room temperature. Following days of drying, they were individually blended into fine powdered materials using a laboratory blender (model 150013, MRC, Durban, South Africa). Exactly, 10 g each of the powdered samples were exhaustively extracted with 400 ml each of ethanol (100%), aq. ethanol (50:50), decoction, and distilled water for 3 days. This was followed by placing the flasks on a horizontal platform shaker (Labcon laboratory Consumables, PTY, Durban, South Africa) at 110 rpm to allow for proper agitation. While, all the extracts were filtered using Whatman No. 1 filter paper, the organic extracts were concentrated on a rotary evaporator under reduced pressure at 40°C (Cole-Parmer, SB 1100, Shanghai, China) and the other aqueous extracts were lyophilized (Virtis Bench Top, SP Scientific Series) to obtain a dry powdered extracts which were used to prepare a stock solution of 1 mg/ml from where other four concentrations were obtained for the in vitro assays. While, the left-over extracts were kept in air-tight container and refrigerated (4°C) for future analysis, the stock of all the extracts was prepared by initial dissolution in 100 μl dimethyl sulfoxide followed by the addition of 900 μl distilled water (to make 1 mg/ml), which was also used for subsequent dilutions.
Chemicals and reagents
Enzymes such as pancreatic α-amylase and rat intestinal α-glucosidase, substrate like para-nitrophenyl-glucopyranoside (pNPG) and starch as well as DPPH, gallic acid, and acarbose were obtained from Sigma–Aldrich (South Africa). While, the water used for the assays was glass-distilled, other chemicals and reagents used were obtained from certified local suppliers in pure analytical form.
Qualitative phytochemical analysis
Various standard methods were employed for the phytochemical analysis of the extracts from L. montana in order to detect the different phytoconstituents (such as alkaloids, anthraquinones, flavonoids, phenols, tannins, triterpenes, and phytosterols) present (Prashant et al., 2011; Sofowara, 2006).
Quantitative phytochemical analysis
Determination of total phenolic content
The method described by Wolfe et al. (2003) was used to determine the total phenolic content in the plant extracts. Briefly, 1 ml aliquot of the extracts was mixed with 5 ml Folin-Ciocalteu reagent (1:10 v/v distilled water) and 4 ml (75 g/L) Na2CO3. The vortexed (15 seconds) tubes were allowed to stand for 30 minutes at 40°C and the absorbance was measured at 765 nm afterward using a microplate reader (BIO-RAD, Model 680, Japan). The equation from the calibration curve of gallic acid was used to extrapolate the total phenolic content and expressed as mg/g gallic acid equivalent.
In vitro antioxidant assays
1, 1-diphenyl-2-picrylhydrazyl radical scavenging assay
The method described by Turkoglu et al. (2007) measures the antioxidant ability of the extract to decolorize the purple-colored methanol solution of DPPH. In brief, 1 ml 0.2 mM DPPH methanol solution was mixed with 1 ml of various concentrations (62–1,000 μg/ml) of the extracts and incubated at 25°C for 30 minutes. Nearly, 300 μl from the mixture was transferred into a 96-well plate in order to measure the absorbance against blank at 516 nm via a microplate reader (BIO-RAD, Model 680, Japan). The percentage inhibition (I%) of the radical by the extract was calculated using the following Equation:
where A control is the absorbance (abs) reading of the control, while A extract represents the abs of the extract. The half maximal concentration (IC50) of the extract that inhibited the activity of DPPH radical was obtained from the dose-response curve using the linear regression equation as follows:
where y was the percentage activity and equals to 50, m was the slope, c was the intercept, and x was the calculated IC50 value.
ABTS radical scavenging ability
The procedure of Re et al. (1999) was employed at determining the scavenging effect of the extract on 2,2-azinobis (3-ethylbenzothiazoline-6) sulphonic acid (ABTS) chromophore which was generated by reacting 7 mM ABTS aqueous solution with K2S2O8 (2.45 mM) in the dark for 16 hours. Approximately, 150 μl extracts (62–1,000 μg/ml) was added to 1.5 ml ABTS+ solution in a test tube and following 15 minutes of incubation at room temperature, the absorbance was measured at 734 nm using a microplate reader.
Nitric oxide scavenging ability
The potential of L. montana extracts to scavenge nitric oxide radical was experimented by adopting the method described by Garrat (1964). Nearly, 200 μl sodium nitroprusside (10 mM) was reacted with 50 μl phosphate buffer saline (pH 7.4) and 50 μl of different concentrations of the extracts (62–1,000 μg/ml) in a 96-well plate for 120 minutes at 25°C. Subsequently, 50 μl was pipetted from the incubated mixtures and mixed with 100 μl sulfanilic acid reagent (0.33% in 20% glacial acetic acid) as well as 100 μl naphthyl ethylenediamine dihydrochloride (0.1% w/v) into another 96-well plate. Following 35 minutes incubation of the whole mixture at 25°C, the absorbance was read at 540 nm. The percentage inhibition and the IC50 calculated [according to Equations (1) and (2) above], respectively.
Reducing power (capacity)
The reducing ability of the extracts was examined based on Oyaizu (1986) method. Aliquots (62–1,000 μg/ml) were added to 1 ml of distilled water, 2.5 ml 0.2 M phosphate buffer (pH 6.6), and 2.5 ml potassium ferrocyanide (1%). Upon subjecting the reacting mixture to 20 minutes incubation at 50°C, 2.5 ml trichloroacetic acid (TCA) was added and centrifuged for 10 minutes at 3,000 rpm. After this, 100 μl from the supernatant was mixed with 100 μl distilled water and 20 μl 0.1% FeCl3 in a 96-well plate which was followed by measuring the absorbance at 700 nm using a microplate reader (BIO-RAD, Model 680, Japan).
Hydroxyl radical inhibitory potential
The assay was done according to the method described by Oboh et al. (2007) which depicts the extracts potential in terminating Fe2+/H2O2-induced deoxyribose decomposition. Nearly, 20 μl of the freshly prepared extracts (62–1,000 μg/ml) was added to 10 μl 20 mM deoxyribose, 40 μl 0.1 M phosphate buffer, 5 μl of 500 μM FeSO4, 50 μl distilled water and incubated at 37°C for 30 minutes. The reaction was halted following the addition of 50 μl of TCA and TBA reagents (2.8% TCA and 0.6% thiobarbituric acid, TBA) and heated (100%) for 20 minutes before the absorbance was read at 532 nm in a microplate reader (BIO-RAD, Model 680, Japan). The percentage inhibition and IC50 were calculated as expressed in Section 2.6.1.
Metal chelating ability
The method of Dinis et al. (1994) was adopted to determine the ability of the extract to chelate metal ions. Briefly, 40 μl of the extract (62–1,000 μg/ml) was added to 80 μl 0.2 mM ferrous chloride solution. The reaction was initiated by the addition of 80 μl of ferrozine (5 mM), followed by incubating the mixture at 25°C for 10 minutes and the absorbance read at 562 nm using a microplate reader (BIO-RAD, Model 680, Japan).
In vitro antidiabetic assays
α-amylase inhibitory and kinetics of inhibition assays
The α-amylase inhibitory activity and kinetics of inhibition were determined using the methods of Elsnoussi et al. (2012) and Kazeem et al. (2013a), respectively. In brief, 50 μl of each of the varying concentration of the extracts (62–1,000 μg/ml) was mixed with 50 μl of 0.02 M sodium phosphate buffer, SPB (pH 6.9) containing 0.5 mg/ml of α-amylase solution. Thereafter, 50 μl of 1% starch solution in 0.02 M SPB (pH 6.9) was added to each test tube at timed intervals to commence the reaction, the whole mixture was incubated for 10 minutes at 25°C and stopped with 100 μl of dinitrosalicylic acid color reagent. Following this, the tubes were suspended in a heated (100°C) water bath for 10 minutes, left to cool at 25°C and diluted with 15 ml distilled water prior reading the absorbance at 504 nm using a spectrophotometer (Biochrom WPA Biowave II, Cambridge, UK). Same procedure was carried out for acarbose used as control and whose values were compared with the extracts. The percentage inhibition and IC50 values were calculated and determined as stated in Section 2.6.1.
The kinetics of α-amylase inhibition by the extracts was determined by reacting 500 μl of the aq.ethanol extract (5 mg/ml) with 500 μl of α-amylase solution (0.5 mg/ml) in a set of tubes and 500 μl of phosphate buffer (0.02 M) with 500 μl of α-amylase solution (0.5 mg/ml) in another set of tubes. Subsequently, varying concentrations (0.3–5.0 mg/ml) of starch as substrate (500 μl) was added to all tubes and the reaction process followed the same procedure as described above. The amount of reducing sugars released was determined spectrophotometrically using maltose standard curve converted to reaction velocities (V). Lineweaver–Burk double reciprocal plot (1/V vs. 1/[S]) was drawn and the mode of α-amylase inhibition by the extract was evaluated (Lineweaver and Burke, 1934).
α-glucosidase inhibitory assay and mode of inhibition
The α-glucosidase inhibitory activity was carried-out according to Elsnoussi et al. (2012) method. Nearly, 50 μl of varying concentrations (62–1,000 μg/ml) of L. montana extracts were mixed with 100 μl of 0.1 M phosphate buffer (pH 6.9) containing 1.0 U/ml of the α-glucosidase solution in 96-well plates. Subsequently, 50 μl of the substrate, 5 mM p-NPG solution in 0.1 M phosphate buffer (pH 6.9) was added to each well at timed intervals and incubated for 15 minutes at 37°C before terminating the process with 50 μl 0.1M sodium carbonate. The absorbance was taken at 405 nm using a microplate reader (BIO-RAD, Model 680, Japan). The percentage inhibition (I%) and IC50 were calculated afterward using Equtations (1) and (2).
The kinetics of α-glucosidase inhibitory activity of L. montana extracts was assessed according to Dnyaneshwar and Archana (2013) method. Briefly, 50 μl of the ethanolic extract was pre-incubated with 100 μl of the α-glucosidase solution in one set of tubes. Concomitantly, 100 μl α-glucosidase was preincubated with 50 μl phosphate buffer (pH 6.9) in another set of five test tubes. 50 μl p-NPG at increasing concentrations (0.63–2.00 mg/ml) was added to both sets of test tubes to initiate the reaction. The reaction mixture was thereafter incubated for 20 minutes at 37°C, and 500 μl of 0.1 M Na2CO3 was introduced to terminate the reaction. The quantity of the reducing sugars formed was spectrophotometrically evaluated using a para-nitrophenol standard curve. Reaction velocities (V) were then determined and the double reciprocal plot of 1/V against 1/[S] was constructed based on the Lineweaver and Burk method to depict the type of inhibition.
Statistical analysis
Statistical analyses were carried out using GraphPad Prism 5 statistical package (GraphPad Software). One-way analysis of variance was used to analyse the data followed by Bonferroni test. Results were expressed as a mean ± standard error of the mean (SEM) of three replicates determination. Statistical significance of their mean values was considered at p < 0.05.
RESULTS
Phytochemical screening
Table 1 depicts the presence of alkaloids, phenols, and triterpenes in all the extracts (pod and seed). Saponins, phytosterols, and tannins were present in all the pod extracts. While, flavonoid was absent in pod and seed extracts, ethanolic extract of the pod and seed, as well as decoction, however, lack anthraquinones. Similarly, aqueous extract of the seed revealed the absence of saponins as well as phytosterol in the ethanolic extract. The highest total phenolic content was observed by the aqueous extracts of the pod (1,511 mg/GAE/ml/extract) and seed (1,506 mg/GAE/ ml/extract) as compared with other extracts (Table 2).
In vitro antioxidant assays
The result from Table 3 indicated that the decoction extract showed the best activity going by half maximal inhibitory concentration (IC50) results in three namely, ABTS (1,371 μg/ml), DPPH (730.40 μg/ml), and hydroxyl radical (798.76 μg/ml) among the six tested radical scavenging assays when compared with other extracts and gallic acids (3,094, 4,125, and 523.70 μg/ml, respectively) which was statistically different (p < 0.05) for the pods. However, aqueous extract of the seed revealed a considerable statistical (p < 0.05) activity in hydroxyl radical (617.80 μg/ml) and metal chelating (59.86 μg/ml) as compared with co-extracts and control. Ethanolic and aqueous ethanolic extracts for both pod and seeds presented a substantial (p < 0.05) nitric oxide scavenging effect and reducing capabilities (Table 4), respectively, in contrast with other extracts though gallic acid activity in the former was superior. Aqueous (Aq.) extract of the pod was a better metal chelator, while the best effect was seen in aqueous ethanolic (1,350 μg/ml) and decoction (791.50 μg/ml) for the ABTS and DPPH respectively when compared with co-extracts and control for the seed of L. montana.
In vitro antidiabetic assays
The aqueous extract (IC50: 338.80 μg/ml) of the pod as well as aq. ethanolic (IC50: 305.70 μg/ml) of the seeds showed the best inhibitory activity (p < 0.05) against α-amylase compared to other extracts and acarbose (p > 0.05). For α-glucosidase, the most significant (p < 0.05) activity was observed with decoction (613.50 μg/ml) for the pod and ethanol (662.90 μg/ml) for the seeds (Table 5). Additionally, the Lineweaver–Burke plot revealing the mode of inhibition of pancreatic α-amylase and intestinal α-glucosidase by aq. ethanolic extract reflected a constant Vmax of 0.92 mM/minutes between the extract and the control with a reduction in Km values from 0.114 to 0.039 (mM)−1 for the pod signifying a competitive α-amylase and non-competitive α-glucosidase arising from constant Km value of 0.18 (mM)−1 (extract and control) with a concomitant reduction in Vmax from 0.105 mM/minutes (extract) to 0.034 mM/minutes (control) (Fig. 1). However, an uncompetitive inhibition by the ethanolic extract of the seed is witnessed for both enzymes as presented in Figure 2 with a reduction from 0.924 to 0.907 mM/minutes for Vmax and 0.14 to 0.03 (mM)−1 Km values.
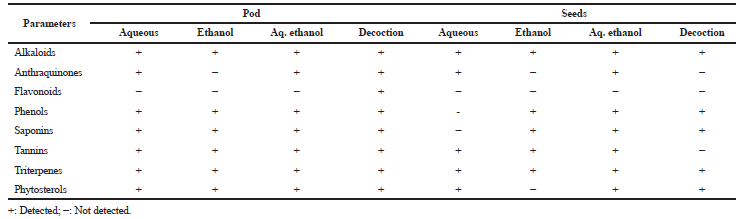 | Table 1. Qualitative phytochemical screening of L. montana pod and seeds extracts. [Click here to view] |
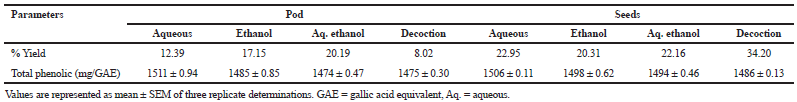 | Table 2. Percentage yield and total phenolic content of L. montana pod and seeds extracts. [Click here to view] |
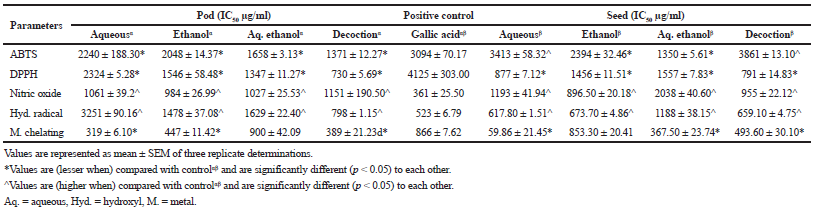 | Table 3. Free radical scavenging potentials of L. montana pod and seeds.Values are represented as mean ± SEM of three replicate determinations. [Click here to view] |
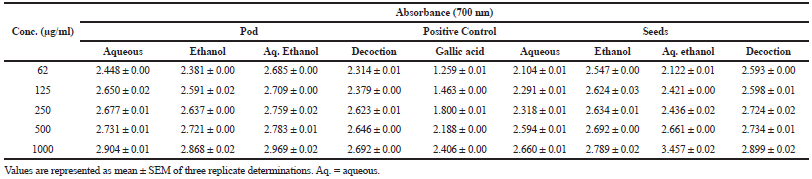 | Table 4. Reducing capabilities of L. montana pod and seeds. [Click here to view] |
 | Table 5. Inhibitory effects of L. montana of pod and seeds extracts on alpha-amylase and alpha-glucosidase activities. [Click here to view] |
DISCUSSION
Medicinal plants (MP) in the recent past are a prominent source of pharmacological and therapeutic agents providing relief or treatment to a number of life-threatening diseases (Firenzuoli and Gori, 2007). These plants are endowed with arrays of important chemical entities such as alkaloids, flavonoids, phenolic, tannins, etc. probably responsible for the bioactivity witnessed in them. Interestingly, some of these secondary metabolites or chemical compounds have been found to be specific or active against numerous health-related disorders (Jamshidi-Kia et al., 2018). The presence of alkaloids, phenols, saponins, triterpenes, and tannins in most of the extracts if not all in this present investigation could suggest the likely antioxidative properties of L. montana since metabolites such as phenols, tannins which are prominent members of polyphenolic compounds are reported to possess free radical scavenging effects (Ayoola et al., 2008; Herrera et al., 2009). Moreover, alkaloids have been established to have diuretic property aside from its ability to lessen the urge for food and water intake (Ojala et al., 2000). Similarly, flavonoids are revealed to have cytotoxic, antimicrobial, antioxidant, antidiabetic, etc. properties (Balogun and Ashafa, 2018; Pandey, 2007; Trifunsch and Ardelean, 2013). All these justify the pharmacological effects of the plant coupled with the reported presence of some of these metabolites in the leaf (Alimi and Ashafa, 2017). In line with the aforementioned, the witnessed highest total phenolic content particularly, with the aqueous extract could be related to the hydroxyl group linked to the aromatic ring conferring on this plant possible free radicals scavenging ability.
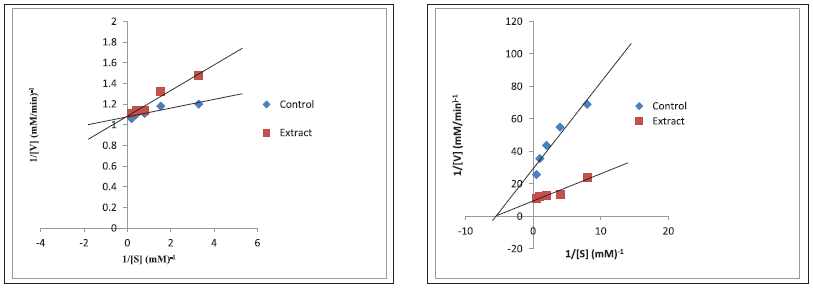 | Figure 1. Competitive inhibition (left) of alpha amylase and non-competitive inhibition of alpha-glucosidase (right) by aqueous-ethanol extract of the L. montana pod. [Click here to view] |
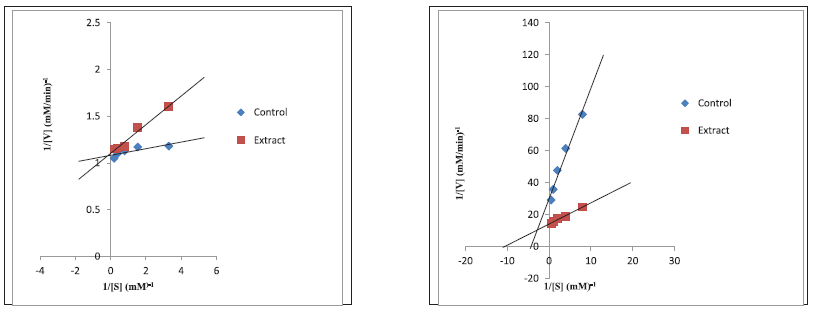 | Figure 2. Uncompetitive inhibitions of alpha amylase (left) and alpha-glucosidase (right) by ethanol extract of the L. montana seed. [Click here to view] |
The serious consequences of free radicals have necessitated an alarming increase in the development of numerous degenerative diseases globally. This effect could be attributed to the imbalance between the body enzymatic or non-enzymatic antioxidants (superoxide dismutase, catalase, glutathione peroxidase, and vitamins C or E) and ROS such as H2O2, OH−, and superoxide anion radicals produced when the body undergoes stress resulting in cell damage and eventual development of ill-effect-associated conditions (Penchant et al., 2004; Steer et al., 2002). Thus the intervention with rich amount of antioxidant particularly from natural origin will to some extent curb the excessive production of free radicals (Balogun and Ashafa, 2016a) since the influence of synthetic antioxidants (butylated hydroxyl anisole, butylated hydroxyl toluene, etc.) have been associated with the possible hepatic damage, cancer development, etc. (Krishnaiah et al., 2011). The present study evaluated the FR scavenging potentials of this plant with the likelihood of providing insight into the pharmacological importance of the herb. The ability of the extract especially decoction (pod) and aqueous (seed) to show better inhibition of the radicals compared to gallic acid is indicative of the radical scavenging capabilities of the plant. However, looking at the report of Alimi and Ashafa (2017), the ethanol extract revealed the best inhibition of most of the radicals showing better effect than the control (Silymarin) in three of the six tested assays. The report of our investigation asides reinforcing the submission of Alimi and Ashafa (2017) on the leaves was in line with the submission of numerous other investigations on the antioxidant potentials of few other MPs such as D. anomala, C. gigantea, G. krebsiana, E. obliqua, Asphalatus linearis, L. frutescens, etc. from our research group and other studies (Balogun and Ashafa, 2016a; 2016b; Gwashu, 2016; Katerere and Eloff, 2005; Mbhele et al., 2015; Sabiu et al., 2017) while suggesting the possibility of natural antioxidants to produce significant activity that could be at par with the synthetic ones as evidenced from the results of the study.
DM is a chronic derangement characterized by hyperglycaemia due to ineffective insulin secretion or both (Khan et al., 2009). In recent times due to side effects from oral hypoglycaemic agents, insight into the use of medicinal plants for the management of diabetes have been recommended by WHO for diabetes control based on their availability, affordability and little or no side effects. Many of these MP such as A. linearis, Artemisia afra, Scelocarya birrea, L. frutescens etc. have been pharmacologically proven to have antidiabetic effect in South Africa (Afolayan and Sunmonu, 2010; Chadwick et al., 2007; Kamakura et al., 2015; Kawano et al., 2009; Mackenzie et al., 2012; Ojewole, 2004; Williams et al., 2013). Interestingly, a prominent measure geared toward diabetes management is via the control of blood glucose level which is partly achieved by inhibiting the carbohydrates-hydrolysing enzymes such as α-amylase and α-glucosidase involved in the continuous digestion of carbohydrates into smaller glucose residues particularly in postprandial hyperglycemia (Balogun and Ashafa, 2017; Gropper and Smith, 2012). In this study, aqueous and decoction extracts of the pod, as well as aq. ethanolic and ethanolic extracts of the seed, showed the strongest inhibition of α-amylase and α-glucosidase respectively going by the respective IC50 values. It is worthy of note that in order for a good hypoglycaemic agent to confer good antidiabetic effect without side effects (such as flatulence, diarrhoea, abdominal discomfort, etc.) similar to those witnessed with synthetic drugs such as acarbose, it should be able to establish mild α-amylase inhibitory (highest IC50 value) and strongest α-glucosidase inhibitory (lowest IC50 value) activities (Kazeem et al., 2016) as witnessed by the ethanolic extract of seed (1,191 and 662.90 μg/ml, respectively) when compared with other extracts. However, it is therefore appropriate to select a particular extract for the pod as a mild α-amylase inhibitor, hence; ethanolic extract (1,815 μg/ml) was chosen. This is hoped that such inhibitors (extracts) could be able to bring the desired result of controlling postprandial hyperglycaemia arising from persistent hydrolysis of starch from dietary foods resulting in the elevated glucose level in the blood. Interestingly, the pod report from this investigation was in consonance with the outcome of Alimi and Ashafa (2017) on the antidiabetic assays where the best inhibition of α-amylase and α-glucosidase were exhibited by aqueous and decoction extracts respectively and this is a pointer to the antihyperglycaemic effect of the plant (Balogun and Ashafa, 2017) although the leaf gave the best result going by the respective IC50 values. Moreover, similar results revealed the excellent inhibition of these enzymes by aqueous extract of S. birrea, Ziziphus mucronata, L. frutescens among others (Da Costa Mousinho, 2013; Elliot, 2011).
The kinetics of enzyme inhibition was incorporated in this study at understanding the mode of inhibition of these enzymes by the extracts. α-amylase was competitively inhibited by aq. ethanolic extract of the pod going by Lineweaver–Burk plot. In competitive inhibition, the inhibitor resembles the substrate and binds exclusively at the active site of the enzyme which means that higher concentration of the inhibitor prevented the substrate binding, hence, kinetically; Vmax is constant while Km value increases, and increasing the substrate concentration could in a way attenuate the inhibition. Relating this mechanism to this study, the active components in the extract competed at the active sites of the enzymes with the substrate (acarbose) and this indicates the likelihood to delay the breakdown of starch to disaccharides (Shai et al., 2010). On the other hand, α-glucosidase was non-competitively inhibited by the same extract. While with non-competitive inhibition, increasing the substrate concentration do not alleviate the inhibition because the inhibitor (active components of the extract) was unable to compete with the substrate but rather binds to free enzymes or at other sites on the enzyme-substrate complex (Zhang et al., 2014), thus raising the substrate concentration (acarbose) is ineffective in attenuating the inhibition, implying that kinetically, Km value would be unchanged but Vmax value of the inhibitor concentration decreases. This suggests that at lower concentration, the inhibitor (extract) could still be effective in slowing down the conversion of disaccharides to sugars (Mogale et al., 2011) in contrast to the greater concentration of acarbose that would be required to exhibit similar effect when large amount of carbohydrates are consumed (Ghadyale et al., 2011). Intriguingly, these types (competitive α-amylase and non-competitive α-glucosidase) of the kinetics of enzyme inhibitions by L. montana pod had been reported in the work of Kazeem et al. (2013b) for M. lucida leaf. Moreover, the uncompetitive inhibition of both α-amylase and α-glucosidase by ethanolic extract of the seed suggest the binding of the active chemical entities to the enzyme-substrate complex which adversely lowers the substrate affinity for the active site, thus ultimately hinders the continuous hydrolysis of oligosaccharides to monosaccharides (Bachhawat, 2011).
Conclusively, it can be inferred going by the results of this investigation that the individual use of pod and seeds could serve as good source of hypoglycaemic agent. The observed effect is reinforced by the antioxidative property of the plant fortified by the rich amount of bioactive chemical constituents as established in the study. Further work is on-going on the isolation and structural elucidation of pharmacologically active compounds that could be responsible for the elicited action.
ACKNOWLEDGMENTS
The authors acknowledge the support from Directorate Research and Development, University of the Free State, South Africa (SA) for the Postdoctoral Research Fellowship granted Dr. FO Balogun (2-459-B3425) tenable at the Phytomedicine and Phytopharmacology Research Group (PpRG), UFS, QwaQwa Campus, South Africa.
CONFLICT OF INTEREST
All the authors declared no conflict of interest.
REFERENCES
Aboyade OM, Styger G, Gibson D, Hughes G. Sutherlandia frutescens: the Meeting of science and traditional knowledge. J Altern Complement Med, 2014; 20(2):71–6.
Alimi AA, Ashafa AOT. An in vitro evaluation of the antioxidant and antidiabetic potential of Sutherlandia montana E. Phillips & R.A. Dyer leaf extracts. Asian Pac J Trop Biomed, 2017; 7(9):765–72.
Alwan A, Maclean D. A review of noncommunicable diseases in low- and middle income countries. Int Health, 2009; 1:3–9.
Anstey KJ, Cherbuin N, Budge M, Young J. Body mass index in midlife and late-life as a risk factor for dementia: a meta-analysis of prospective studies. Obesity Rev, 2011; 12:e426–37.
Ayoola GA, Coker HAB, Adesegun SA, Adepoju-Bello AA, Obaweya K, Ezennia EC, Atangbayila TO. Phytochemical screening and antioxidant activities of some selected medicinal plants used for malarial therapy in Southwestern Nigeria. Trop J Pharm Res, 2008; 7(3):1019–24.
Bachhawat JA, Shihabudeen MS, Thirumurugan K. Screening of Fifteen Indian ayurvedic plants for alpha-glucosidase inhibitory activity and enzyme kinetics. Int J Pharm Pharm Sci, 2011; 4:267–74.
Balogun FO, Ashafa AOT. Antioxidant and hepatoprotective activities of aqueous root extracts of Dicoma anomala (Sond.) against carbon tetrachloride-induced liver damage in rats. J Trad Chin Med, 2016a; 36(4):505–13.
Balogun FO, Ashafa AOT. Antioxidant, hepatoprotective and ameliorative potentials of aqueous leaf extract of Gazania krebsiana (Less.) against carbon tetrachloride (CCl4)-induced liver injury in Wistar rats. Trans R Soc S Afr, 2016b; 71(2):145–56.
Balogun FO, Ashafa AOT. Aqueous roots extract of Dicoma anomala (Sond.) extenuates postprandial hyperglycaemia in vitro and its modulation against on the activities of carbohydrate-metabolizing enzymes in streptozotocin-induced diabetic Wistar rats. S Afr J Bot, 2017; 112:102–11.
Balogun FO, Ashafa AOT. Cytotoxic, kinetics of inhibition of carbohydrate-hydrolysing enzymes and oxidative stress mitigation by flavonoids roots extract of Dicoma anomala (Sond.). Asian Pac J Trop Med, 2018; 11(1):24–31.
Chadwick WA, Roux S, van de Venter M, Louw J, Oelofsen W. Anti-diabetic effects of Sutherlandia frutescens in Wistar rats fed a diabetogenic diet. J Ethnopharmacol, 2007; 109:121–7.
Da Costa Mousinho NMH. In vitro assessment of the antidiabetic activity of Sclerocarya birrea and Ziziphus mucronata [Online]. MSc dissertation, University of Pretoria, 2018. Available via https://repository.up.ac.za/bitstream/handle/2263/33337/DaCostaMousinh_InVitro_2013.pdf?sequence=1 (Accessed 7April 2018).
Dinis TCP, Madeira VMC, Almeida MLM. Action of phenolic derivates (acetaminophen, salycilate and 5-aminosalycilate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch Biochem Biophys, 1994; 315:161–9.
Diogo CV, Suski JM, Lebiedzinska M, Karkucinska-Wieckowska A, Wojtala A, Pronicki M, Duszynski J, Pinton P, Portincasa P, Oliveira PJ, Wieckowski MR. Cardiac mitochondrial dysfunction during hyperglycemia: the role of oxidative stress and p66Shc signaling. Int J Biochem Cell Biol, 2013; 45(1):114–22.
Dnyaneshwar MN, Archana RJ. In vitro inhibitory effects of Pithecellobium dulce (Roxb.) Benth. seeds on intestinal a-glucosidase and pancreatic a-amylase. J Biochem Technol, 2013; 4(3):616–21.
Elliot GP. Implementation of novel flow cytometric methods to assess the in vitro antidiabetic mechanism of a Sutherlandia frutescens extract. Ph.D. Thesis, Nelson Mandela Metropolitan University, 2011.
Elsnoussi AHM, Mohammad JAS, Lee FA, Amirin S, Sue HC, Soo CT. Potent α-glucosidase and α-amylase inhibitory activities of standardized 50% ethanolic extracts and sinensetin from Orthosiphon stamineus Benth as anti-diabetic mechanism. BMC Complement Altern Med, 2012; 12:176.
Firenzuoli F, Gori L. Herbal medicine today: clinical and research issues. Evid Based Complement Altern Med, 2007; 4(Suppl 1):37–40.
Folli F, Corradi D, Fanti P, Davalli A, Paez A, Giaccari A, Perego C, Muscogiuri G. The role of oxidative stress in the pathogenesis of type 2 diabetes mellitus micro- and macrovascular complications: avenues for a mechanistic-based therapeutic approach. Curr Diabetes Rev, 2011; 7(5):313–24.
Garrat DC. The quantitative analysis of drugs. Chapman and Hall, Japan, p 456, 1964.
Ghadyale V, Takalikar S, Haldavnekar V, Arvindekar A. Effective control of postprandial glucose level through inhibition of intestinal alpha-glucosidase by Cymbopogon martinii (Roxb.). Evid Based Complement Altern Med, 2011; 2012:1–6.
Goldblatt P, Manning J. Cape plants. A conspectus of the Cape Flora of South Africa. Strelitzia 9. National Botanical Institute, Pretoria, 2000.
Gropper SS, Smith JL. Advanced nutrition and human metabolism. Cengage Learning, Hampshire, UK, 2012.
Gwashu A. Protective properties of rooibos (Aspalathus linearis) flavonoids on the prevention of skin cancer [Online]. MSc dissertation, Stellenbosch University, Available via file:///C:/Users/ufs/Downloads/gwashu_protective_2016.pdf (Accessed 6 April 2016).
Herrera EA, Verkerk MM, Derks JB, Giussani DA. Antioxidant treatment alters peripheral vascular dysfunction induced by postnatal glucocorticoid therapy in rats. PLoS One, 2010; 5:e9250.
Jamshidi-Kia F, Lorigooini Z, Amini-Khoei H. Medicinal plants: past history and future perspective. J Herbmed Pharmacol, 2018; 7(1):1–7.
Kamakura R, Jin Son M, de Beer D, Joubert E, Miura Y, Yagasak K. Antidiabetic effect of green rooibos (Aspalathus linearis) extract in cultured cells and type 2 diabetic model KK-Ay mice. Cytotechnol, 2015; 67:699–710.
Katerere DR, Eloff JN. Antibacterial and antioxidant activity of Sutherlandia frutescens (Fabaceae), a reputed anti-HIV/AIDS phytomedicine. Phytother Res, 2005; 19:779–81.
Kawano A, Nakamura H, Hata S, Minakawa M, Miura Y, Yagasaki K. Hypoglycemic effect of Aspalathin, a rooibos tea component from Aspalathus linearis, in type 2 diabetic model db/db mice. Phytomed, 2009; 16(5):437–43.
Kazeem MI, Mayaki AM, Ogungbe BF, Ojekale AB. In-vitro studies on Calotropis procera leaf extracts as inhibitors of key enzymes linked to diabetes mellitus. Iranian J Pharm Res, 2016; 15(Special issue):37–44.
Kazeem MI, Abimbola SG, Ashafa AOT. Inhibitory potential of Gossypium arboreum Linn leaf extracts on diabetes key enzymes (α-amylase and α-glucosidase). Bangladesh J Pharmacol, 2013a; 8:149–55.
Kazeem MI, Adamson JO, Ogunwande IO. Modes of inhibition of α-amylase and α-glucosidase by aqueous extract of Morinda lucida Benth Leaf. BioMed Res Int, 2013b; Article ID 527570:6; doi:10.1155/2013/527570
Khan A, Zaman G, Anderson RA. Bay leaves improve glucose and lipid profile of people with type 2 diabetes. J Clin Biochem Nutr, 2009; 44:52–6.
Kitabchi AE, Umpierrez GE, Miles JM, Fisher JN. Hyperglycemic crises in adult patients with diabetes. Diabetes Care, 2009; 32(7):1335–43.
Krishnaiah D, Rosalam S, Rajesh N. A review of the antioxidant potential of medicinal plant species. Food Bioprod Process, 2011; 89:217–33.
Lineweaver H, Burke D. The determination of enzyme dissociation constants. J Am Chem Soc, 1934; 56:658–66.
Lobo V, Patil A, Phatak A, Chandra N. Free radicals, antioxidants and functional foods: impact on human health. Pharmacogn Rev, 2010; 4(8):118–26.
Mackenzie J, Koekemoer T, Roux S, van de Venter M, Dealtry GB. Effects of Sutherlandia frutescens on the lipid metabolism in an insulin resistant rat model and 3T3-L1 adipocytes. Phytother Res, 2012; 26:1830–7.
Mbhele N, Balogun FO, Kazeem MI, Ashafa AOT. In vitro studies on the antimicrobial, antioxidant and antidiabetic potential of Cephalaria gigantea. Bangladesh J Pharmacol, 2015; 10:214–21.
Moffett R. Sesotho plant and animal names and plants used by the Basotho. Sun Media, Bloemfontein, p 302, 2010.
Mogale AM, Lebelo LS, Thovhogi N, de Freitas AN, Shai LJ. α-amylase and α-glucosidase inhibitory effects of Sclerocarya birrea [(A. Rich.) Hochst.] subspecies caffra (Sond) Kokwaro (Anacardiaceae) stem-bark extracts. Afr J Biotechnol, 2011; 10(66):15033–9.
Nepal B, Brown LJ, Anstey KJ. Rising midlife obesity will worsen future prevalence of dementia. PLoS One, 2014; 9(9):e99305; doi:10.1371/journal. pone.0099305
Oboh G, Puntel RL, Rocha JBT. Hot pepper (Capsicum annuum, Tepin and Capsicum chinese, Habanero) prevents Fe2+ induced lipid peroxidation in brain in vitro. Food Chem, 2007; 102:178–85.
Ojala T, Remes S, Haansuu P, Vuorela H, Hiltunen R, Haahtela K. Antimicrobial activity of some coumarin containing herbal plants growing in Finland. J Ethnopharmacol, 2000; 73:299–305.
Ojewole JA. Analgesic, antiinflammatory and hypoglycemic effects of Sutherlandia frutescens R. BR. (variety Incana E. MEY.) [Fabaceae] shoot aqueous extract. Methods Find Exp Clin Pharmacol, 2004; 26(6):409.
Oyaizu M. Studies on products of Browning reaction: antioxidative activities of product of Browning reaction prepared from glucosamine. Jpn J Nutr, 1986; 44:307–15.
Pandey AK. Anti-staphylococcal activity of a pan-tropical aggressive and obnoxious weed Parihenium histerophorus: an in vitro study. Nat Acad Sci Lett, 2007; 30(11–12):383–6.
Peuchant E, Brun J, Rigalleau V, Dubourg L, Thomas M, Daniel J. Oxidative and antioxidative status in pregnant women with either gestational or type 1 Diabetes. Clin Biochem, 2004; 37:293–8.
Prashant T, Bimlesh K, Mandeep K, Gurpreet K, Harleen K. Phytochemical screening and extraction: a review. Int J Pharm Sci, 2011; 1(1):98–106.
Re R, Pellegrini N, Proteggente A. Antioxidant activity applying an improved ABTS radical cation decolourisation assay. Free Rad Biol Med, 1999; 26(9–10):1231–7.
Rother KI. Diabetes treatment—bridging the divide. New Engl J Med, 2007; 356(15):1499–501.
Sabiu S, Ajani EO, Sunmonu TO, Balogun FO, Ashafa AOT, Othman BR, Olowa KS. Mechanism of hepatoprotective potential of aqueous leaves extract of Eucalyptus obliqua (Myrtaceae) in carbon tetrachloride intoxicated Wistar rats. Appl J Pharm Sci, 2017; 7(8):183–90.
Shai LJ, Masoko P, Mokgotho MP, Magano SR, Mogale AM, Boaduo N, Eloff JN. Yeast alpha glucosidase inhibitory and antioxidant activities of six medicinal plants collected in Phalaborwa, South Africa. S Afr J Bot, 2010; 76(3):465–70.
Shoback D. Chapter 17. Greenspan’s basic & clinical endocrinology. 9th edition, McGraw-Hill Medical, New York, pp 197–214, 2011.
Sofowara A. Medical plants and traditional medicine in Africa. Spectrum Books Ltd, Ibadan, pp 191–289, 2006.
Steer P, Milligard J, Sarabi DM, Wessby B, Kahan T. Cardiac and vascular structure and function are related to lipid peroxidation and metabolism. Lipids, 2002; 37:231–6.
Sunmonu TO, Afolayan AJ. Protective effect of Artemisia afra Jacq. on Isoproterenol-Induced Myocardial Injury in Wistar Rats. Food Chem Toxicol, 2010; 48:1969–72. http://dx.doi.org/10.1016/j.fct.2010.04.046
Trifunsch IT, Ardelean DG. Flavonoid extraction from Ficus carica leaves using different techniques and solvents. J Nat Sci, 2013; 125:79–84.
Turkoglu A, Duru ME, Mercan N, Kivrak I, Gezer K. Antioxidant and antimicrobial activities of Laetiporus sulphureus (bull). Food Chem, 2007; 101:267–73.
van Wyk BE, Albrecht C. A review of the taxonomy, ethnobotany, chemistry and pharmacology of Sutherlandia frutescens (Fabaceae). J Ethnopharmacol, 2008; 119:620–9.
van Wyk BE, Gericke N. People’s plants. A guide to useful plants of southern Africa. Briza Publications, Pretoria, South Africa, 2000.
Willi C, Bodenmann P, Ghali WA, Faris PD, Cornuz J. Active smoking and the risk of type 2 diabetes: a systematic review and meta-analysis. JAMA, 2007; 298(22):2654–64.
Williams S, Roux S, Koekemoer T, van de Venter M, Dealtry G. Sutherlandia frutescens prevents changes in diabetes-related gene expression in a fructose-induced insulin resistant cell model. J Ethnopharmacol, 2013; 146:482–9.
Wolfe K, Wu X, Liu RH. Antioxidant activity of apple peels. J Agric Food Chem, 2003; 51:609–14.
World Health Organization (WHO). Diabetes [Online]. Available via http://www.who.int/news-room/fact-sheets/detail/diabetes (Accessed 6 February 2018).
World Health Organisation (WHO). Diabetes fact sheet N 312 [Online]. Available via http://www.who.int/mediacentre/factsheets/fs312/en/ (Accessed 6 February 2018).
World Health Organization. Dept. of Non-communicable Disease Surveillance. Definition, diagnosis and classification of diabetes mellitus and its complications: report of a WHO consultation. Part 1, Diagnosis and classification of diabetes mellitus [Online]. World Health Organization, Geneva, Switzerland. Available via http://www.who.int/iris/handle/10665/66040. (Accessed 6 February 2018).
Yang H, Jin X, Kei Lam CW, Yan SK. Oxidative stress and diabetes mellitus. Clin Chem Lab Med, 2011; 49(11):1773–82.
Young IS, Woodside JV. Antioxidants in health and disease. J Clin Pathol, 2001; 54:176–86.
Zhang H, Wang G, Dong J. Inhibitory properties of aqueous ethanol extracts of propolis on alpha-glucosidase. Evid Based Complement Altern Med, 2015; 2015:1–8.